| Wenker synthesis | |
|---|---|
| Named after | Henry Wenker |
| Reaction type | Ring forming reaction |
| Identifiers | |
| Organic Chemistry Portal | wenker-synthesis |
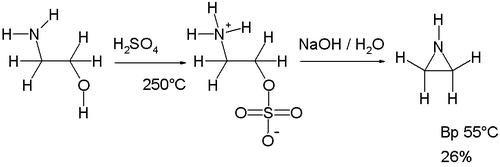
The Wenker synthesis is an organic reaction converting a beta amino alcohol to an aziridine with the help of sulfuric acid. It is used industrially for the synthesis of aziridine itself.[1]
The original Wenker synthesis of aziridine itself takes place in two steps. In the first step ethanolamine is reacted with sulfuric acid at high temperatures (250 °C) to form the sulfate monoester. This salt is then reacted with sodium hydroxide in the second step forming aziridine. The base abstracts an amine proton enabling it to displace the sulfate group. A modification of this reaction involving lower reaction temperatures (140–180 °C) and therefore reduced charring increases the yield of the intermediate.[2]
The Wenker synthesis protocol using trans-2-aminocyclooctanol, available from reaction of ammonia with the epoxide of cyclooctene, gives a mixture of cyclooctenimine (the Wenker aziridine product) and cyclooctanone (a competing Hofmann elimination product).[3]
Further reading
- Wenker, Henry (1935). "The Preparation of Ethylene Imine from Monoethanolamine". Journal of the American Chemical Society. 57 (1): 2328–28. doi:10.1021/ja01314a504.
References
- ^ Steuerle, Ulrich; Feuerhake, Robert (2006). "Aziridines". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a03_239.pub2.
- ^ Leighton, Philip A.; Perkins, William A.; Renquist, Melvin L. (1947). "A Modification of Wenker's Method of Preparing Ethyleneimine". J. Am. Chem. Soc. 69 (6): 1540. doi:10.1021/ja01198a512.
- ^ Kashelikar, D. V.; Fanta, Paul E. (1960). "Chemistry of Ethylenimine. VII. Cyclooctenimine or 9-Azabicyclo[6.1.0]nonane". J. Am. Chem. Soc. 82 (18): 4927–30. doi:10.1021/ja01503a044.
![9-Azabicyclo[6.1.0]nonane synthesis](http://img2.tfd.com/wiki/thumbs/8/81/500px-WenkerSynthApplic.png)