 | |
| Names | |
|---|---|
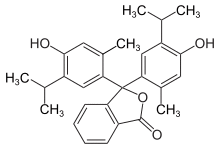
| IUPAC name 3,3-bis(4-hydroxy-2-methyl-5-propan-2-ylphenyl)-2-benzofuran-1-one | |
| Identifiers | |
3D model (JSmol) | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.004.300 |
| EC Number |
|
PubChem CID | |
| UNII | |
CompTox Dashboard (EPA) | |
| |
SMILES
| |
| Properties | |
Chemical formula | C28H30O4 |
| Molar mass | 430.544 g·mol−1 |
| Appearance | White powder |
| Melting point | 248 to 252 °C (478 to 486 °F; 521 to 525 K) (decomposes) |
| Hazards | |
| R-phrases (outdated) | 4, 10 |
| S-phrases (outdated) | S22 S24/25 |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Thymolphthalein is a phthalein dye used as an acid–base (pH) indicator. Its transition range is around pH 9.3–10.5. Below this pH, it is colorless; above, it is blue. The molar extinction coefficient for the blue thymolphthalein dianion is 38,000 M−1 cm−1 at 595 nm.[1]
| Thymolphthalein (pH indicator) | ||
| below pH 9.3 | above pH 10.5 | |
| 9.3 | ⇌ | 10.5 |
| Thymolphthalein (pH indicator) | ||
| below pH <0 | above pH 9.3 | |
| <0 | ⇌ | 9.3 |
Thymolphthalein is also known to have use as a laxative [2] and for disappearing ink.[3]
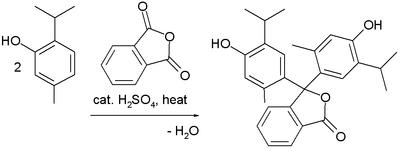
Preparation
Thymolphthalein can be synthesized from thymol and phthalic anhydride
References
- ^ Hahn HH; Cheuk SF; Elfenbein S; Wood WB (April 1970). "Studies on the Pathogenesis of Fever: Xix. Localization of Pyrogen in Granulocytes". J. Exp. Med. 131 (4): 701–9. doi:10.1084/jem.131.4.701. PMC 2138774. PMID 5430784.
- ^ Hubacher, MH; Doernberg, S; Horner, A (1953). "Laxatives: chemical structure and potency of phthaleins and hydroxyanthraquinones". J Am Pharm Assoc Am Pharm Assoc. 42 (1): 23–30. doi:10.1002/jps.3030420108. PMID 13034620.
- ^ Katz, David A. (1982). "Disappearing Ink" (PDF). www.chymist.com. Retrieved August 14, 2017.