 | |||
| | |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name Pyridazine[1] | |||
| Systematic IUPAC name 1,2-Diazabenzene | |||
| Other names 1,2-Diazine Orthodiazine Oizine | |||
| Identifiers | |||
3D model (JSmol) | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.005.478 | ||
| EC Number |
| ||
PubChem CID | |||
| UNII | |||
CompTox Dashboard (EPA) | |||
InChI
| |||
SMILES
| |||
| Properties | |||
Chemical formula | C4H4N2 | ||
| Molar mass | 80.090 g·mol−1 | ||
| Appearance | Colorless liquid | ||
| Density | 1.107 g/cm3 | ||
| Melting point | −8 °C (18 °F; 265 K) | ||
| Boiling point | 208 °C (406 °F; 481 K) | ||
| miscible | |||
| Solubility | miscible in dioxane, ethanol soluble in benzene, diethyl ether negligible in cyclohexane, ligroin | ||
Refractive index (nD) | 1.52311 (23.5 °C) | ||
| Thermochemistry | |||
Std enthalpy of formation (ΔfH⦵298) | 224.9 kJ/mol | ||
| Hazards | |||
| Flash point | 85 °C (185 °F; 358 K) | ||
| Related compounds | |||
Related compounds | pyridine, pyrimidine, pyrazine | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
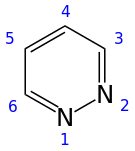
Pyridazine is a heterocyclic organic compound with the molecular formula (CH)4N2. It contains a six-membered ring with two adjacent nitrogen atoms, and is aromatic.[2] It is a colorless liquid with a boiling point of 208 °C. It is isomeric with two other (CH)4N2 rings, pyrimidine and pyrazine.
Occurrence
Pyridazines are rare in nature, possibly reflecting the scarcity of naturally occurring hydrazines, common building blocks for the synthesis of these heterocycles. The pyridazine structure is a popular pharmacophore which is found within a number of herbicides such as credazine, pyridafol and pyridate. It is also found within the structure of several drugs such as cefozopran, cadralazine, minaprine, pipofezine, and hydralazine.
Synthesis
In the course of his classic investigation on the Fischer indole synthesis, Emil Fischer prepared the first pyridazine via the condensation of phenylhydrazine and levulinic acid.[3] The parent heterocycle was first prepared by oxidation of benzocinnoline to the pyridazinetetracarboxylic acid followed by decarboxylation. A better route to this otherwise esoteric compound starts with the maleic hydrazide. These heterocycles are often prepared via condensation of 1,4-diketones or 4-ketoacids with hydrazines.[4]
References
- ^ "Front Matter". Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 141. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ^ Gumus, S. (2011). "A computational study on substituted diazabenzenes" (PDF). Turk J Chem. 35: 803–808. Archived from the original (PDF) on 2016-03-03. Retrieved 2014-04-10.
- ^ Fischer, E. (1886). "Indole aus Phenylhydrazin". Justus Liebigs Annalen der Chemie. 236 (1–2): 126–151. doi:10.1002/jlac.18862360107.
- ^ Tišler, M.; Stanovnik, B. (1968). "Pyridazines". Advances in Heterocyclic Chemistry. 9: 211–320. doi:10.1016/S0065-2725(08)60374-8. ISBN 9780120206094.