 | |
| Names | |
|---|---|
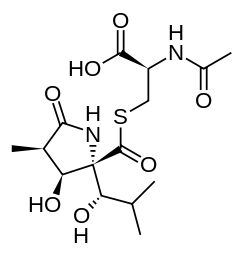
| IUPAC name 2-(acetylamino)-3-[({3-hydroxy-2-[1-hydroxy-2-methylpropyl]-4-methyl-5-oxopyrrolidin-2-yl}carbonyl)sulfanyl]propanoic acid | |
| Identifiers | |
3D model (JSmol) | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| MeSH | Lactacystin |
PubChem CID | |
CompTox Dashboard (EPA) | |
| |
SMILES
| |
| Properties | |
Chemical formula | C15H24N2O7S |
| Molar mass | 376.42 g·mol−1 |
| log P | 0.086 |
| Acidity (pKa) | 3.106 |
| Basicity (pKb) | 10.891 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Lactacystin is an organic compound naturally synthesized by bacteria of the genus Streptomyces first identified as an inducer of neuritogenesis in neuroblastoma cells in 1991.[1] The target of lactacystin was subsequently found to be the proteasome on the basis of its affinity for certain catalytic subunits of the proteasome by Fenteany and co-workers in 1995.[2] The proteasome is a protein complex responsible for the bulk of proteolysis in the cell, as well as proteolytic activation of certain protein substrates. Lactacystin was the first non-peptidic proteasome inhibitor discovered and is widely used as a research tool in biochemistry and cell biology. The transformation product of lactacystin clasto-lactacystin β-lactone (also known as omuralide) covalently modifies the amino-terminal threonine of specific catalytic subunits of the proteasome, a discovery that helped to establish the proteasome as a mechanistically novel class of protease: an amino-terminal threonine protease. The molecule is commonly used as in biochemistry and cell biology laboratories as a selective inhibitor of the proteasome.[2][3] The first total synthesis of lactacystin was developed in 1992 by Corey and Reichard,[4] and a number of other syntheses of this molecule have also been published. There are more than 1,660 entries for lactacystin in PubMed as of January 2019.
See also
- Satoshi Ōmura
References
- ^ Omura S, Fujimoto T, Otoguro K, Matsuzaki K, Moriguchi R, Tanaka H, Sasaki Y. (1991). Lactacystin, a novel microbial metabolite, induces neuritogenesis of neuroblastoma cells: S. Omura, et al. J. Antibiot. 44(1):113-6.
- ^ a b Fenteany G, Standaert RF, Lane WS, Choi S, Corey EJ, Schreiber SL (1995). "Inhibition of proteasome activities and subunit-specific amino-terminal threonine modification by lactacystin". Science. 268 (5211): 726–31. Bibcode:1995Sci...268..726F. doi:10.1126/science.7732382. PMID 7732382. S2CID 37779687.
- ^ Fenteany G, Schreiber SL (1998). "Lactacystin, proteasome function, and cell fate". J. Biol. Chem. 273 (15): 8545–8. doi:10.1074/jbc.273.15.8545. PMID 9535824.
- ^ "Total Synthesis of Lactacystin" Corey, E. J.; Reichard, G. A. J. Am. Chem. Soc. 1992, 114, 10677.