Benzo[c]cinnoline 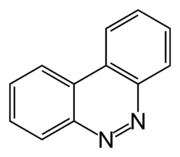 |
| Names |
| IUPAC name Benzo[c]cinnoline |
| Other names Diphenylenazone; phenazone; 9,10-diazaphenanthrene; 2,2'-azobiphenyl; 3,4-benzocinnoline; 5,6-phenanthroline |
| Identifiers |
| | |
3D model (JSmol) | |
| ChemSpider | |
| | |
| | |
InChI=1S/C12H8N2/c1-3-7-11-9(5-1)10-6-2-4-8-12(10)14-13-11/h1-8H  Key: SWJXWSAKHXBQSY-UHFFFAOYSA-N  InChI=1/C12H8N2/c1-3-7-11-9(5-1)10-6-2-4-8-12(10)14-13-11/h1-8H Key: SWJXWSAKHXBQSY-UHFFFAOYAY
|
SMILES C1=CC=C2C(=C1)C3=CC=CC=C3N=N2
|
| Properties |
| | C12H8N2 |
| Molar mass | 180.21 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). |
| | |
Benzo[c]cinnoline is a tricyclic organic compound with the formula C12H8N2. Formally this species is derived by oxidative dehydrogenation of 2,2'-diaminobiphenyl. This heterocycle reacts with iron carbonyls to form C12H8N2Fe2(CO)6.[1]
See also
References
- ^ R. P. Bennett, "Iron Carbonyl Complexes of Azo Compounds" Inorganic Chemistry, Volume 9, pp. 2184-6 (1970) (description of the first iron carbonyl derivative)
This article is copied from an
article on Wikipedia® - the free encyclopedia created and edited by its online user community. The text was not checked or edited by anyone on our staff. Although the vast majority of Wikipedia® encyclopedia articles provide accurate and timely information, please do not assume the accuracy of any particular article. This article is distributed under the terms of
GNU Free Documentation License.
All content on this website, including dictionary, thesaurus, literature, geography, and other reference data is for informational purposes only. This information should not be considered complete, up to date, and is not intended to be used in place of a visit, consultation, or advice of a legal, medical, or any other professional.
