 | |
 | |
| Names | |
|---|---|
| IUPAC name 4-[9-(4-hydroxy-2-methyl-5-propan- 2-yl-phenyl)-7,7-dioxo-8-oxa- 7λ6-thiabicyclo[4.3.0]nona-1,3,5-trien-9-yl]- 5-methyl-2-propan-2-yl-phenol | |
| Other names α-hydroxy-α,α-bis(5-hydroxycarvacryl)- o-toluenesulfonic acid γ-sultone; thymolsulfonephthalein | |
| Identifiers | |
3D model (JSmol) | |
| ChemSpider | |
| ECHA InfoCard | 100.000.886 |
PubChem CID | |
| UNII | |
CompTox Dashboard (EPA) | |
| |
SMILES
| |
| Properties | |
Chemical formula | C27H30O5S |
| Molar mass | 466.59 g·mol−1 |
| Appearance | Brownish-green crystal powder |
| Melting point | 221–224 °C (430–435 °F; 494–497 K) decomposes[1] |
| Insoluble | |
| UV-vis (λmax) | 594 nm (1st) 376 nm (2nd)[1] |
| Hazards | |
| Main hazards | Harmful |
EU classification (DSD) (outdated) | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Thymol blue (thymolsulfonephthalein) is a brownish-green or reddish-brown crystalline powder that is used as a pH indicator. It is insoluble in water but soluble in alcohol and dilute alkali solutions.
| Thymol blue (pH indicator) | ||
| below pH 8.0 | above pH 9.6 | |
| 8.0 | ⇌ | 9.6 |
| Thymol blue (pH indicator) | ||
| below pH 1.2 | above pH 2.8 | |
| 1.2 | ⇌ | 2.8 |
It transitions from red to yellow at pH 1.2–2.8 and from yellow to blue at pH 8.0–9.6. It is usually a component of Universal indicator.
At wavelength (378 - 382) nm, extinction coefficient > 8000 and at wavelength (298 - 302) nm , the extinction coefficient > 12000.[2]
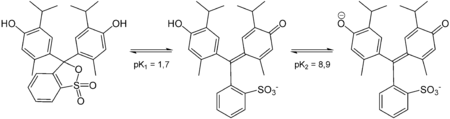
Structures
Thymol blue has different structures at different pH.
Safety
It may cause irritation. Its toxicological properties have not been fully investigated. Harmful if swallowed, Acute Toxicity. Only Hazardous when percent values are above 10%.[3]
Bibliography
- Merck. "Thymol Blue." The Merck Index. 14th ed. 2006. Accessed via web on 2007-02-25.
References
- ^ a b Thymol Blue
- ^ "Product Specification: Thymol Blue- ACS reagent" (PDF). Sigma-Aldrich. 9 October 2017. p. 1.
- ^ "Thymol blue". PubChem. Retrieved 2017-10-09.