 | |
| Clinical data | |
|---|---|
| Routes of administration | By mouth |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
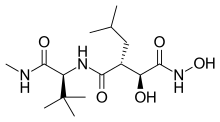
| Formula | C15H29N3O5 |
| Molar mass | 331.413 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| (verify) | |
Marimastat was a proposed antineoplastic drug developed by British Biotech. It acted as a broad-spectrum matrix metalloproteinase inhibitor.[1][2]
Marimastat performed poorly in clinical trials,[3] and development was terminated.
See also
- Batimastat
References
- ^ "Marimastat". National Cancer Institute.
- ^ Millar AW, Brown PD, Moore J, Galloway WA, Cornish AG, Lenehan TJ, Lynch KP (January 1998). "Results of single and repeat dose studies of the oral matrix metalloproteinase inhibitor marimastat in healthy male volunteers". British Journal of Clinical Pharmacology. 45 (1): 21–6. doi:10.1046/j.1365-2125.1998.00639.x. PMC 1873993. PMID 9489589.
- ^ Sparano JA, Bernardo P, Stephenson P, Gradishar WJ, Ingle JN, Zucker S, Davidson NE (December 2004). "Randomized phase III trial of marimastat versus placebo in patients with metastatic breast cancer who have responding or stable disease after first-line chemotherapy: Eastern Cooperative Oncology Group trial E2196". Journal of Clinical Oncology. 22 (23): 4683–90. doi:10.1200/JCO.2004.08.054. PMID 15570070.