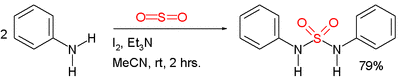 | |
 | |
| Names | |
|---|---|
| IUPAC name Sulfuric diamide | |
| Other names Sulphamide Sulfuryl amide | |
| Identifiers | |
3D model (JSmol) | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.029.330 |
PubChem CID | |
| UNII | |
CompTox Dashboard (EPA) | |
InChI
| |
SMILES
| |
| Properties | |
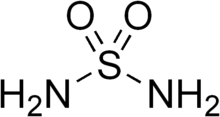
Chemical formula | H4N2O2S |
| Molar mass | 96.11 g/mol |
| Appearance | White orthorhombic plates |
| Melting point | 93 °C (199 °F; 366 K) |
| Boiling point | 250 °C (482 °F; 523 K) (decomposes) |
| Freely soluble | |
| -44.4·10−6 cm3/mol | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Sulfamide (IUPAC name: sulfuric diamide) is a chemical compound with the molecular structure H2NSO2NH2. Sulfamide is produced by the reaction of sulfuryl chloride with ammonia. Sulfamide was first prepared in 1838 by the French chemist Henri Victor Regnault.[2]
Sulfamide functional group
In organic chemistry, the term sulfamide may also refer to the functional group which consists of at least one organic group attached to a nitrogen atom of sulfamide.
Symmetric sulfamides can be prepared directly from amines, sulfur dioxide gas and an oxidant:[3]
In this example, the reactants are aniline, triethylamine, and iodine. Sulfur dioxide is believed to be activated through a series of intermediates: Et3N-I+-I−, Et3N-I+-I3− and Et3N+-SO2−.
The sulfamide functional group is an increasingly common structural feature used in medicinal chemistry.[4]
See also
References
- ^ Merck Index, 11th Edition, 8894.
- ^ Regnault, Victor (1838) "Sur l'acide chlorosulfurique et la sulfamide" (On sulfuryl chloride and sulfamide), Annales de chimie et de physique, series 2, 69 : 170-184; see especially "Action de gaz ammoniac sec sur la liqueur chlorosulfurique" (Action of dry ammonia gas on liquid sulfuryl chloride), pages 176-180.
- ^ Leontiev, A. V.; Dias, H. V. R.; Rudkevich, D. M. (2006). "Sulfamides and sulfamide polymers directly from sulfur dioxide". Chemical Communications. 2006 (27): 2887–2889. doi:10.1039/b605063h.
- ^ Reitz, A. B.; Smith, G. R.; Parker, M. H. (2009). "The Role of Sulfamide Derivatives in Medicinal Chemistry: A Patent Review (2006 – 2008)". Expert Opinion on Therapeutic Patents. 19 (10): 1449–1453. doi:10.1517/13543770903185920. PMID 19650745.