 | |
| Identifiers | |
|---|---|
3D model (JSmol) | |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.162.252 |
IUPHAR/BPS | |
| MeSH | Cyclic+ADP-Ribose |
PubChem CID | |
| UNII | |
CompTox Dashboard (EPA) | |
| |
SMILES
| |
| Properties | |
| C15H21N5O13P2 | |
| Molar mass | 541.301 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
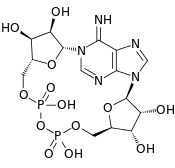
Cyclic ADP Ribose, frequently abbreviated as cADPR, is a cyclic adenine nucleotide (like cAMP) with two phosphate groups present on 5' OH of the adenosine (like ADP), further connected to another ribose at the 5' position, which, in turn, closes the cycle by glycosidic bonding to the nitrogen 1 (N1) of the same adenine base (whose position N9 has the glycosidic bond to the other ribose).[1][2] The N1-glycosidic bond to adenine is what distinguishes cADPR from ADP-ribose (ADPR), the non-cyclic analog. cADPR is produced from nicotinamide adenine dinucleotide (NAD+) by ADP-ribosyl cyclases (EC 3.2.2.5) as part of a second messenger system.
Function
cADPR is a cellular messenger for calcium signaling.[3] It stimulates calcium-induced calcium release at lower cytosolic concentrations of Ca2+. Primary target of cADPR is the endoplasmic reticulum Ca2+ uptake mechanism. cADPR mobilizes Ca2+ from the endoplasmic reticulum by activation of ryanodine receptors,[4] a critical step in muscle contraction.[5]
cADPR also acts as an agonist for the TRPM2 channel. but less potently than ADPR.[6] cADPR and ADPR act synergistically, with both molecules enhancing the action of the other molecule in activating the TRPM2 channel.[7]
Potentiation of Ca2+ release by cADPR is mediated by increased accumulation of Ca2+ in the sarcoplasmic reticulum.[8]
Metabolism
cADPR and ADPR are synthesized from NAD+ by the bifunctional ectoenzymes of the CD38 family (also includes the GPI-anchored CD157 and the specific, monofunctional ADP ribosyl cyclase of the mollusc Aplysia).[9][10][11] The same enzymes are also capable of hydrolyzing cADPR to ADPR. Catalysis proceeds via a covalently bound intermediate. The hydrolysis reaction is inhibited by ATP, and cADPR may accumulate. Synthesis and degradation of cADPR by enzymes of the CD38 family involve, respectively, the formation and the hydrolysis of the N1-glycosidic bond. In 2009, the first enzyme able to hydrolyze the phosphoanhydride linkage of cADPR, i.e. the one between the two phosphate groups, was reported.[12]
The SARM1 enzyme also catalyzes the formation of cADPR from NAD+.[13]
See also
- NAADP
- IP3
- ADP-ribose
References
- ^ Lee HC, Walseth TF, Bratt GT, Hayes RN, Clapper DL (1989). "Structural determination of a cyclic metabolite of NAD+ with intracellular Ca2+-mobilizing activity". J. Biol. Chem. 264 (3): 1608–15. PMID 2912976.
- ^ Lee HC, Aarhus R, Levitt D (1994). "The crystal structure of cyclic ADP-ribose". Nat. Struct. Biol. 1 (3): 143–4. doi:10.1038/nsb0394-143. PMID 7656029. S2CID 9049850.
- ^ Guse AH (2004). "Regulation of calcium signaling by the second messenger cyclic adenosine diphosphoribose (cADPR)". Curr. Mol. Med. 4 (3): 239–48. doi:10.2174/1566524043360771. PMID 15101682.
- ^ Galione A, Chuang K (2020). "Pyridine Nucleotide Metabolites and Calcium Release from Intracellular Stores". Advances in Experimental Medicine and Biology. 1131: 371–394. doi:10.1007/978-3-030-12457-1_15. ISBN 978-3-030-12456-4. PMID 31646518.
- ^ Santulli G, Marks AR (2015). "Essential Roles of Intracellular Calcium Release Channels in Muscle, Brain, Metabolism, and Aging". Current Molecular Pharmacology. 8 (2): 206–22. doi:10.2174/1874467208666150507105105. PMID 25966694.
- ^ Yu P, Cai X, Liang Y, Yang W (2019). "Roles of NAD + and Its Metabolites Regulated Calcium Channels in Cancer". Molecules. 25 (20): 4826. doi:10.3390/molecules25204826. PMC 7587972. PMID 33092205.
- ^ Lee HC (2011). "Cyclic ADP-ribose and NAADP: fraternal twin messengers for calcium signaling". Science China Life Sciences. 54 (8): 699–711. doi:10.1007/s11427-011-4197-3. PMID 21786193. S2CID 24286381.
- ^ Lukyanenko, V; Györke, I; Wiesner, T. F.; Györke, S (2001). "Potentiation of Ca(2+) release by cADP-ribose in the heart is mediated by enhanced SR Ca(2+) uptake into the sarcoplasmic reticulum". Circulation Research. 89 (7): 614–22. doi:10.1161/hh1901.098066. PMID 11577027.
- ^ Prasad GS, McRee DE, Stura EA, Levitt DG, Lee HC, Stout CD (1996). "Crystal structure of Aplysia ADP-ribosyl cyclase, a homolog of the bifunctional ectozyme CD38". Nat. Struct. Biol. 3 (11): 957–64. doi:10.1038/nsb1196-957. PMID 8901875. S2CID 21978229.
- ^ Liu Q, Kriksunov IA, Graeff R, Munshi C, Lee HC, Hao Q (2005). "Crystal structure of the human CD38 extracellular domain". Structure. 13 (9): 1331–9. doi:10.1016/j.str.2005.05.012. PMID 16154090.
- ^ Guse AH (2004). "Biochemistry, biology, and pharmacology of cyclic adenosine diphosphoribose (cADPR)". Curr. Med. Chem. 11 (7): 847–55. doi:10.2174/0929867043455602. PMID 15078169.
- ^ Canales J, Fernández A, Rodrigues JR, Ferreira R, Ribeiro JM, Cabezas A, Costas MJ, Cameselle JC (2009). "Hydrolysis of the phosphoanhydride linkage of cyclic ADP-ribose by the Mn2+-dependent ADP-ribose/CDP-alcohol pyrophosphatase". FEBS Lett. 583 (10): 1593–8. doi:10.1016/j.febslet.2009.04.023. hdl:10400.8/3028. PMID 19379742. S2CID 28571921.
- ^ Lee HC, Zhao YJ (2019). "Resolving the topological enigma in Ca 2+ signaling by cyclic ADP-ribose and NAADP". Journal of Biological Chemistry. 294 (52): 19831–19843. doi:10.1074/jbc.REV119.009635. PMC 6937575. PMID 31672920.